|
The Phase III programme will investigate two doses of ‘719 (125mcg and 62.5mcg) and ‘719/VI (125/25mcg and 62.5/25mcg) across the six further studies, which will all commence within the next quarter. The study initiated today is a 52-week randomised study to evaluate the long term safety and tolerability of ‘719 (125mcg) alone, as well as the combination with ‘719/VI (125/25mcg). These molecules act through two mechanisms: antagonism of acetylcholine muscarinic receptors and agonism of beta 2 adrenoreceptors.The medicine will be administered using a new dry powder inhalation device. ‘719/VI combines two bronchodilator molecules currently under development - ‘719, a long-acting muscarinic antagonist (LAMA) and VI, a long-acting beta agonist (LABA). Relovair is a once-daily inhaled corticosteroid (ICS)/long-acting beta-agonist (LABA) combination treatment, currently under development, comprising fluticasone furoate and vilanterol (FF/VI). The second is the start of an extensive study of 16,000 patients to assess the potential for Relovair to improve survival in those with COPD and a history of, or at risk from, cardiovascular disease. The programme will also include two further studies assessing the effect of ‘719/VI on exercise endurance. Patients are now being enrolled in a large safety study, which will be followed shortly by four large pivotal studies that will compare improvements in lung function between ‘719/VI, its components, placebo and tiotropium. The first is the initiation of the Phase III programme for the once-daily LAMA/LABA dual bronchodilator GSK573719/vilanterol (‘719/VI), which will evaluate over 5,000 patients globally. (NASDAQ: THRX) today announced major milestones in two clinical development programmes focused on new treatments for patients with chronic obstructive pulmonary disease (COPD), a leading cause of chronic illness and death worldwide. GlaxoSmithKline (GSK) and Theravance, Inc. 
Issued: London UK and South San Francisco, CA Pragmatic trials powered to detect real-life differences in HRQoL and head-to-head comparison are needed to guide clinical practice in terms of PROs.Relovair™ programme expanded by start of large Phase IIIb COPD outcomes study Moreover, due to the different molecule properties, treatment schedule, and device characteristics of each FDC, a generalized judgment seems inappropriate. Conclusion: LABA/LAMA FDCs may be helpful in improving HRQoL, but because of the heterogeneity of performed trials, strong conclusions cannot be drawn. In 5 out of 6 RCTs having LABA/ICS as comparators, LABA/LAMA FDC had a similar effect and only 1 showed significant improvement in HRQL compared to LABA/ICS FDC. LABA/LAMA FDCs significantly improved HRQoL versus placebo in 9 out of 11 trials, while change when compared to other LABA or LAMA monocomponents was significantly better in 11 out 24. Using the St George’ s Respiratory Questionnaire, 27 out of 28 trials assessed HRQoL. 
Results: Twenty-eight RCTs ( n = 32, 165 COPD patients) investigating the impact of fixed LABA/LAMA combinations on HRQoL were included. 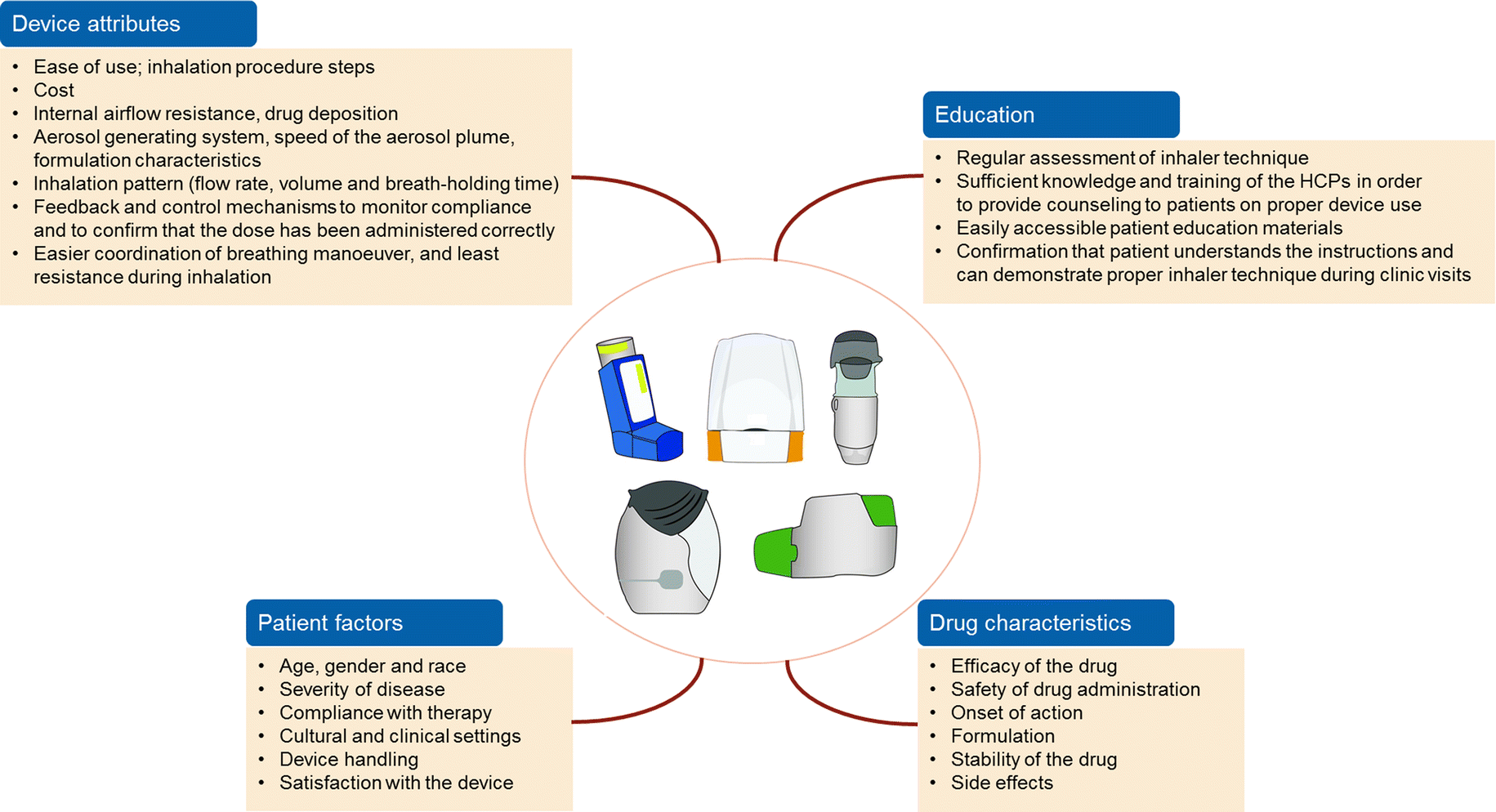
Methods: A systematic literature search for randomized controlled trials (RCTs) about the impact of LABA/LAMA FDCs versus placebo, LABA or LAMA or LABA/ICS on HRQoL in Chronic obstructive pulmonary disease (COPD) patients has been performed. Objectives: To review current available evidence about the treatment effect of fixed LABA/LAMA FDCs on HRQoL. Background: While fixed dose combinations (FDCs) of long-acting beta 2-agonists (LABAs) and long-acting muscarinic antagonists (LAMAs) are increasingly tested on their efficacy in improving lung function, their effectiveness on Patient Reported Outcomes (PROs) such as Health Related Quality of Life (HRQoL) and Health Status (HS) deserve more attention.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
